At Applied Catalysts, we understand the monumental importance of catalysts in accelerating and...

Case Studies in Commercializing Catalytic Chemical Technology
This article explores real-world case studies that demonstrate how Applied Catalysts have helped companies navigate the complexities of bringing new catalytic processes to market.
The original presentation was presented at the 2025 SOCMA show in Nashville, Tennessee.
Case Study #1: Doubling Destruction Rates in Indoor Air Treatment
Industry: Air Purification
Challenge: Develop a catalyst capable of eliminating a proprietary indoor air pollutant with improved efficiency over existing materials.
Our Approach:
Design a bifunctional catalyst to increase the rates of adsorption and destruction.
- Material A: Promoters are added that favor the adsorption of a strongly bound intermediate.
- Material B: Material structure is modified in the presence of promoters to further increase destruction rates.
Samples were tested using a commercial recirculation unit and a custom build 30m³ chamber. The catalyst modified using real-time performance metrics.
Results:
The final catalyst achieved a 2X increase in destruction rate compared to the current state-of-the-art control material. The new formulation was sufficient to provide several market advantages.
This collaboration demonstrates how targeted catalyst formulation, rooted in fundamental principles, can deliver measurable commercial benefits.
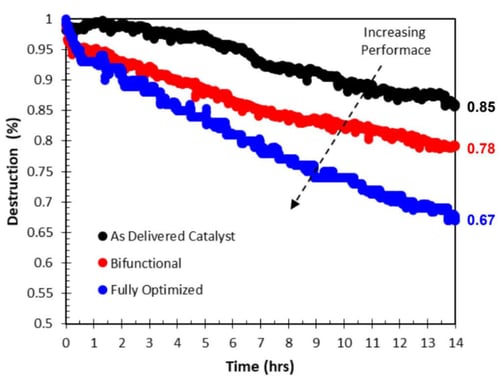
Figure 1: Destruction rates for two next generation air purification catalysts (Red + Blue) compared to the current state-of-the-art control (Black).
Case Study #2: Redesigning a Partial Oxidation Reactor with Tubular Geometry
Industry: Chemicals
Challenge: Improve efficiency and throughput in a partial oxidation process.
Our Approach:
Applied Catalysts designed a custom jacketed tubular reactor using Python modeling. The model included:
- Mole and energy balances
- Rate law expressions for catalyst performance over the range of operating conditions.
- Radial and axial temperature and conversion profiling.
- Momentum balance to account for pressure drop through the bed.
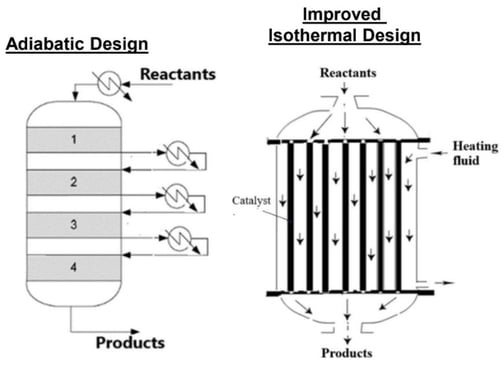
Figure 2: Example adiabatic reactor (left) compared to the target tubular isothermal design (right).
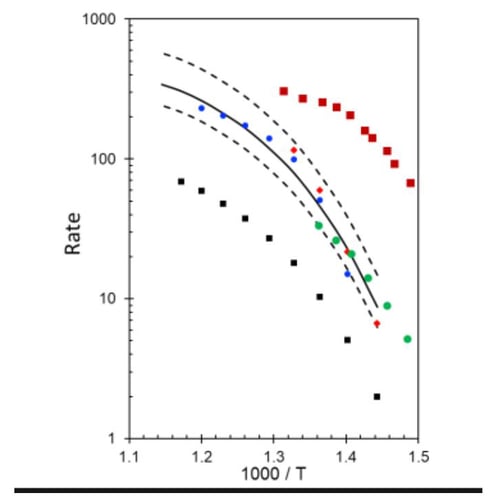
Figure 2: Kinetic data for several catalyst formulations (colored dots) compared to a modeled expression (solid black). A sensitivity analysis was run to explore results based on variation.
Results:
Modeling successfully predicted greater than 95% reactant conversion within a 30ft jacketed tube. The exotherm was successfully managed with axial heat removal along the tube length.
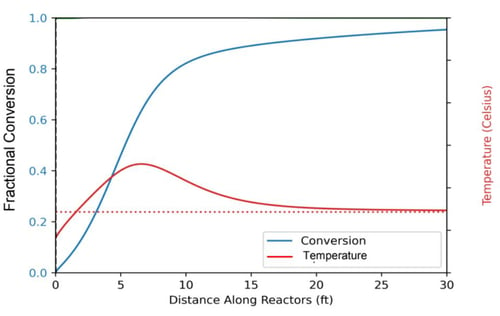
Figure 3: Fractional conversion and temperature as a function of distance along the reactor bed.
This configuration offers several process advantages including:
- The option to significantly increase single pass conversion by operating at higher pressures in tubes.
- Boost steam recovery by capturing the exotherm in a thermal fluid rather than atmospheric losses.
- Preheat the reactor to reduce startup times for operational agility.
This case highlights how advanced reactor engineering and catalyst modeling are essential to modern catalyst manufacturing and process integration.
Case Study #3: Commercial Hydrogenation of Proprietary Oleochemicals
Industry: Oleochemicals
Challenge: Improve the process economics in the hydrogenation of two proprietary feedstocks originating from a palm oil intermediate or a sucrose core modified oil.
Batch Process Issues:
- 10–15% product loss due to filtration inefficiencies
- High catalyst attrition rates
- Slow throughput due to multi-step processing
Our Solution:
Develop a continuous fixed bed process using an 11-ft length, 1.25 inch ID single shell-and-tube pilot reactor. Catalysts and process parameters were screened under industrial heat and mass transfer conditions to accurately determine scaled production rates.
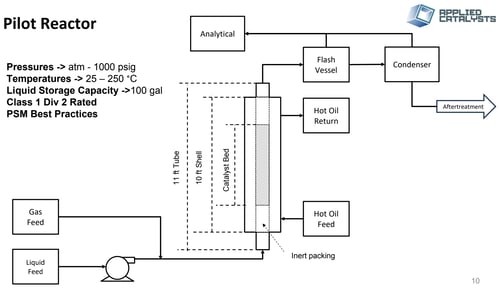
Results:
Catalysts were loaded with identical bed volumes and tested at the temperature and pressure used in batch configuration. Oil flow rate was then varied to observe changes in single pass conversion, Figure 4. Palm oil conversion was significantly lower than the sucrose core oil and experienced a classic Langmuir type curve indicating external mass transfer limitation.
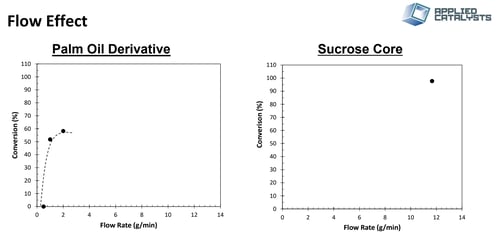
Figure 4: Single pass conversion for a palm oil derived oleochemical (left) and sucrose core oleochemical (right) as a function of flow rate. The beds were loaded with identical catalyst volume and operated at similar pressures.
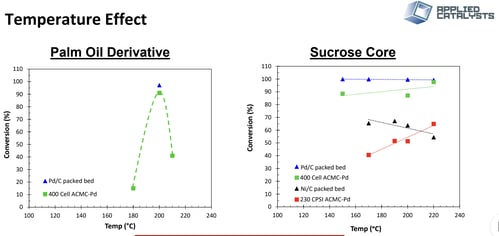
Temperature and catalyst studies were conducted to increase single pass rates, Figure 5. Four (4) catalysts were tested including a palladium (Pd) supported on granular carbon, nickel (Ni) supported on granular carbon, Pd supported on 400 CPSI carbon monolith, and Pd supported on 230 CPSI carbon monolith.
The main finding was that the system is hydrogen starved and externally mass transfer limited. This is evident in the palm oil data where rates of saturation decrease with increasing reaction temperature indicating a competing mechanism to Arrhenius kinetics. This behavior was attributed to decreased hydrogen solubility at higher temperatures leading to lower saturation rates.
A comparison of Granular Ni/C and Pd/C in sucrose core saturation indicates a flat trend with increasing temperature for Pd/C where conversion decreases with temperature for Ni/C. It is well established that the rate of hydrogen dissociation on Ni is significantly less than on Pd. Therefore, a hydrogen starved surface would be more apparent for a Ni based system due to decreased concentrations of monoatomic hydrogen.
Commercial Scale-Up Recommendations:
Further optimization was performed to determine an ideal set of operating conditions. These conditions were used to size a commercial fixed bed reactor with an annual production capacity of 4,500 metric tons. The ease of saturation for different Oleochemicals is apparent with the sucrose core oil requiring 40, 10-ft, tubes (131 ft^3 of catalyst) and the palm oil derivative requiring 5,500, 30ft tubes (1290 ft^3 of catalyst). A batch system was recommended for the palm oil system and a continuous system for the sucrose core oil.
This study highlights the importance of evaluating chemistry with reactor studies as part of a commercialization strategy for specific oleochemicals. Our recommendations enabled our client to choose the optimal configuration for each feedstock, improving process performance and reducing capital investment risk.
Partner with Applied Catalysts
Applied Catalysts specializes in overcoming the challenges of commercial scaleup. By leveraging our diverse expertise in new process development and existing manufacturing assets for catalyst manufacturing at all scales our team can help bring your technology to market.
If you're evaluating new catalytic chemical technology or struggling to scale one, you need a partner who can think strategically, act quickly, and deliver. Reach out to Applied Catalysts on your next project need.


